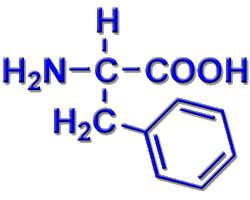
Glycine, the major amino acid found in gelatin, was named for its sweet taste (Greek glykys, meaning “sweet”). It was obtained from protein found in asparagus juice (hence the name). Chemical Properties: Aromatic (Aromatic R-group), Physical Properties: Nonpolar As the name suggests, phenylalanine, an essential amino acid, is a derivative. Due to its hydrophobicity L-phenylalanine is nearly always found. Do coinage metal anions interact with substituted benzene derivatives. Phenylalanine and tryptophan contain phenyl and indole rings and are classified as non-polar. Zahra Aliakbar Tehrani, Zahra Jamshidi, Hossein Farhangian. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of Figure 22.4. The Ionic Hydrogen/Deuterium Bonds between Diammoniumalkane Dications and Halide Anions. The first amino acid to be isolated was asparagine in 1806. a) The alpha helix, beta pleated sheet and beta turns are examples of protein secondary structure. Ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. The only amino acid whose R group has a pK a (6.0) near physiological pHĪlmost as strong a base as sodium hydroxide Oxidation of two cysteine molecules yields cystineĪmino acids with a negatively charged R groupĬarboxyl groups are ionized at physiological pH also known as aspartateĬarboxyl groups are ionized at physiological pH also known as glutamateĪmino acids with a positively charged R group Named for its similarity to the sugar threose Side chain functions as a methyl group donorĬontains a secondary amine group referred to as an α-imino acidĪmino acids with a polar but neutral R group The only amino acid lacking a chiral carbonĪn essential amino acid because most animals cannot synthesize branched-chain amino acidsĪlso classified as an aromatic amino acid \): Common Amino Acids Found in Proteins Common Name
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
